Content Warning – This was a post written for my friends on Facebook who have been discussing testing a great deal; this is a non-referenced post written on a tube train which may or may not be of interest to the wider world
I’m on a tube so this will, again, be stream of consciousness, but I’ve seen multiple conversations about testing in recent days and so here are a few comments/thoughts:
Testing is undertaken for 2 separate, but linked, reasons:
- 1 – epidemiological testing in order to control transmission risk and instigate additional measures such as isolation.
- 2 – clinical management so we know what your viral load (we’ll come back to this term) is doing and how you may be responding to medication.
The testing that is being done for SARS CoV2 testing in the community is called Pillar 2 testing. The testing in hospitals is called Pillar 1 testing. Most of the testing undertaken in hospital labs is logically much more focused on aim 2 rather than aim 1, although we care about aim for prevention of hospital outbreaks. The main function of Pillar 2 testing is based around aim 1, but it also acts to give information if you present at a hospital. Clinical management is not the main aim of Pillar 2: most of your healthcare management will be based on symptoms, irrespective of a positive result, and on physiological measurements such as 02 Stats.
Back to how testing works.
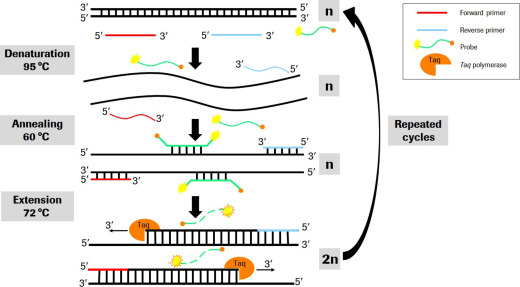
The gold standard test for SARS CoV2 is polymerase chain reaction or PCR, where we look for fragments of the virus, and then create replicates of this original fragment until we have enough to detect. This means that if you have more virus on board you make the number of replicates needed to be detected as positive quicker than someone who is shedding only a low level of the virus.
We normally deal with positive tests in terms of cycle threshold (CT). This is the number of replication cycles required to detect the virus. If your CT value is very low i.e. 20 cycles then you had millions of viral particles present in your sample. If your CT is 38, you had hundreds and it took a lot longer to replicate enough to detect. Still with me? As that was the technical bit! The thing to take away is (counter intuitively) a low CT = lots of virus, a high CT = low levels of virus. This is important because every test has a limit to its sensitivity. PCR can detect down to a few hundred or few thousand copies of the virus but it has its limits.
One of the problems with SARS CoV2 is where the virus initially does most of its replication, i.e. in the nasopharynx: the back of your throat and upper nose. That means to try and get a good sample to enable the testing you need to get into there which is not only uncomfortable but pretty hard to do to yourself in terms of visualisation. This means that, even though the test process itself is pretty good, the samples we put into it are often not that well taken (which is why in hospitals they are taken by someone else) and so you may not have enough virus present to count as positive when the PCR is run. This brings me onto the picture below and asymptomatic testing. The first thing I want you to remember is that a test is only valid at the moment it is taken. It does not (if negative) represent what will happen 5 minutes or an hour later. Therefore taking a screen when you are asymptomatic has very limited value to either of our aims. Within healthcare and for contact tracing purposes, we default to the fact that you could be asymptomatically shedding virus for 48 hours before you develop symptoms but that brings us onto viral load. This is called the pre-symptomatic phase.
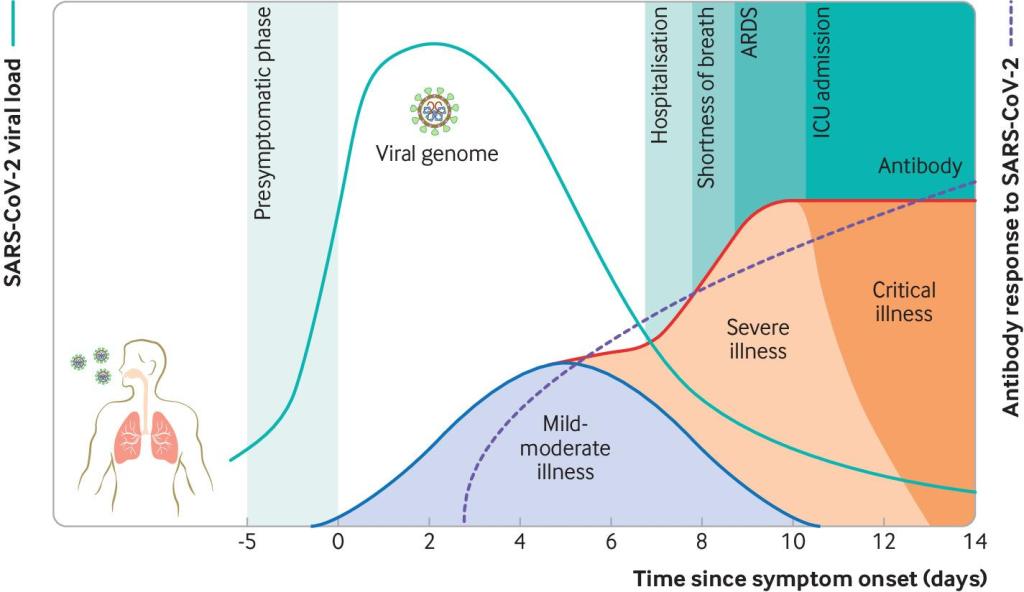
Viral load is a term we use to talk about how much virus you are shedding or have in your cells. The viral load gradually increases in the asymptomatic phase of infection BUT, and there is a BUT, not everyone will shed virus as the same level even when symptomatic. Some people will control the virus better in terms of replication and will therefore not be detected positive in the pre-symptomatic phase or even on day one of symptoms. The most sensitive day to test is actually on day three after symptom onset. Therefore an early negative test is not helpful. Yes, if positive it means you can put your actions earlier but it is not reliable. Therefore asymptomatic testing needs to always be undertaken with caution and is only valuable in very specific settings. The reason we talked so much about asymptomatic transmission earlier in the pandemic is that we didn’t have our symptoms right. We were looking for flu-like symptoms and ignoring things like anosmia (loss of taste and smell) and, now we’re including it, we don’t see much that meets asymptomatic transmission outside of the 48 hours before symptoms develop. As time goes on replication can predominately move into the lower respiratory tract, i.e. lungs etc. and then you may get negative nose and throat swabs where as deeper samples taken from the lungs are positive.
What does this mean?
- If you are asymptomatic please don’t request a test as it probably doesn’t give you the information you think you are getting.
- If someone has been asked to isolate as a contact that doesn’t mean they have exposed you, it means we are asking them to isolate to reduce the risk of them exposing you in the pre-symptom 48-hour phase and so there is no alerting necessary until they have symptoms.
- If you’ve been exposed to a positive test: work on the isolation guidelines, based on symptoms not just on the test results.
- Samples can be taken in a way that doesn’t capture the true picture: they may be taken too early and/or be below test sensitivity Pillar 2 testing aims to support stopping transmission and not clinical management, so bear that in mind
Right, back to the coal face
All opinions on this blog are my own